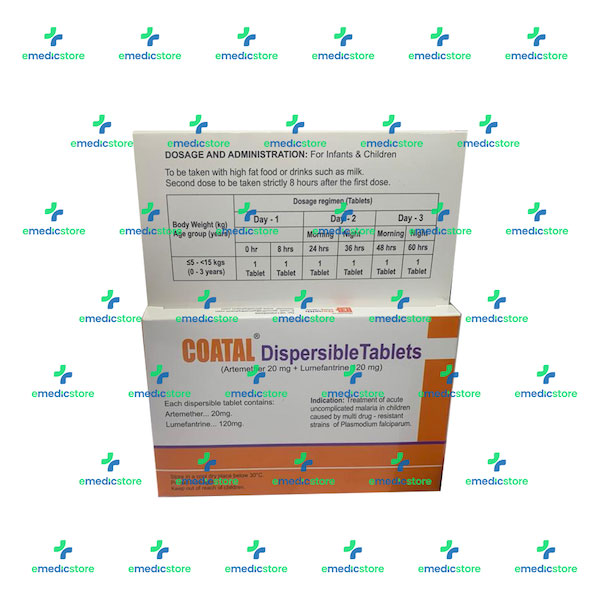
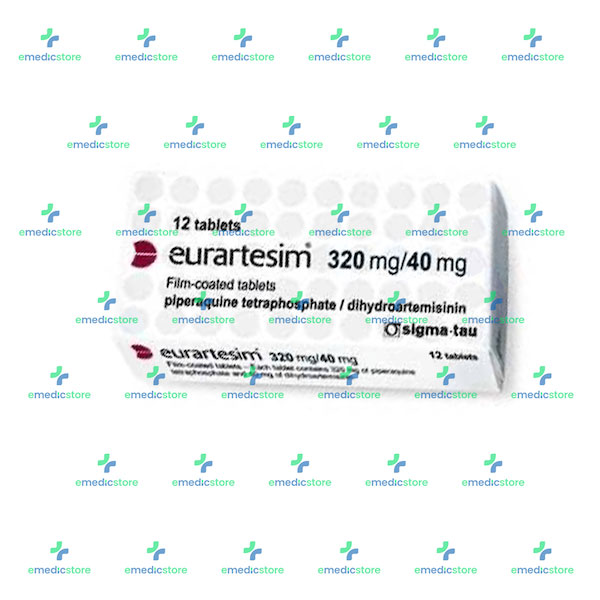
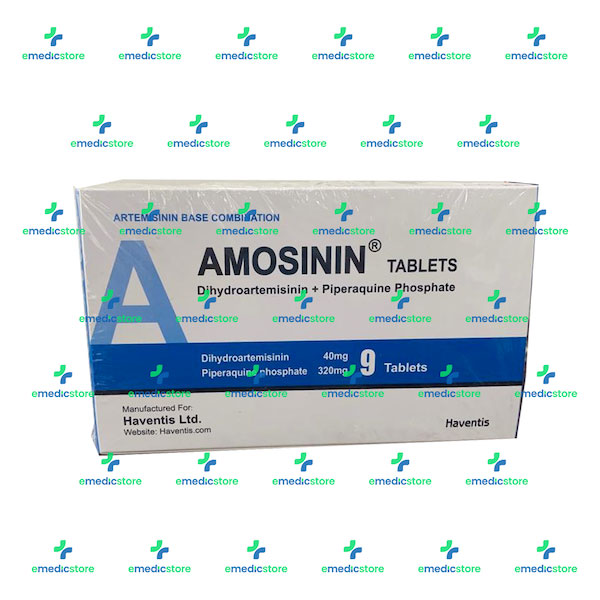
Stability of Dihydroartemisinin–Piperaquine Tablet Halves during Prolonged Storage under Tropical Conditions

Efficacy and Tolerability Outcomes of a Phase II, Randomized, Open-Label, Multicenter Study of a New Water-Dispersible Pediatric Formulation of Dihydroartemisinin-Piperaquine for the Treatment of Uncomplicated Plasmodium falciparum Malaria in African ...